<p style="text-align:justify"><font size="3"> Plasma medicine is an emerging field combining plasma physics and engineering, medicine, bioengineering to use plasmas for therapeutic applications. MpNL investigates the effect of cold atmospheric plasma (CAP) on cancer therapy.</font> </p><!-- [et_pb_line_break_holder] --><br><!-- [et_pb_line_break_holder] --><p style="text-align:justify"><font size="3">There is convincing evidence that CAP selectively causes cancer cell death. MpNL has previously investigated the application of CAP in cancer therapy. This includes the role of reactive species (reactive oxygen and nitrogen), cell cycle modification, in vivo application, interaction of cancer cells with gold nanoparticles, and computational oncology.</font></p><!-- [et_pb_line_break_holder] --><br><!-- [et_pb_line_break_holder] --><p style="text-align:justify"><font size="3">Current projects include: physics of atmospheric plasma jet, discharges in media, self-organization of atmospheric discharges, plasma-stimulated media, mechanism of plasma selectivity, plasma application for cancer treatment in vivo, pre-clinical studies, and nanoparticle-cell interactions.</font></p>
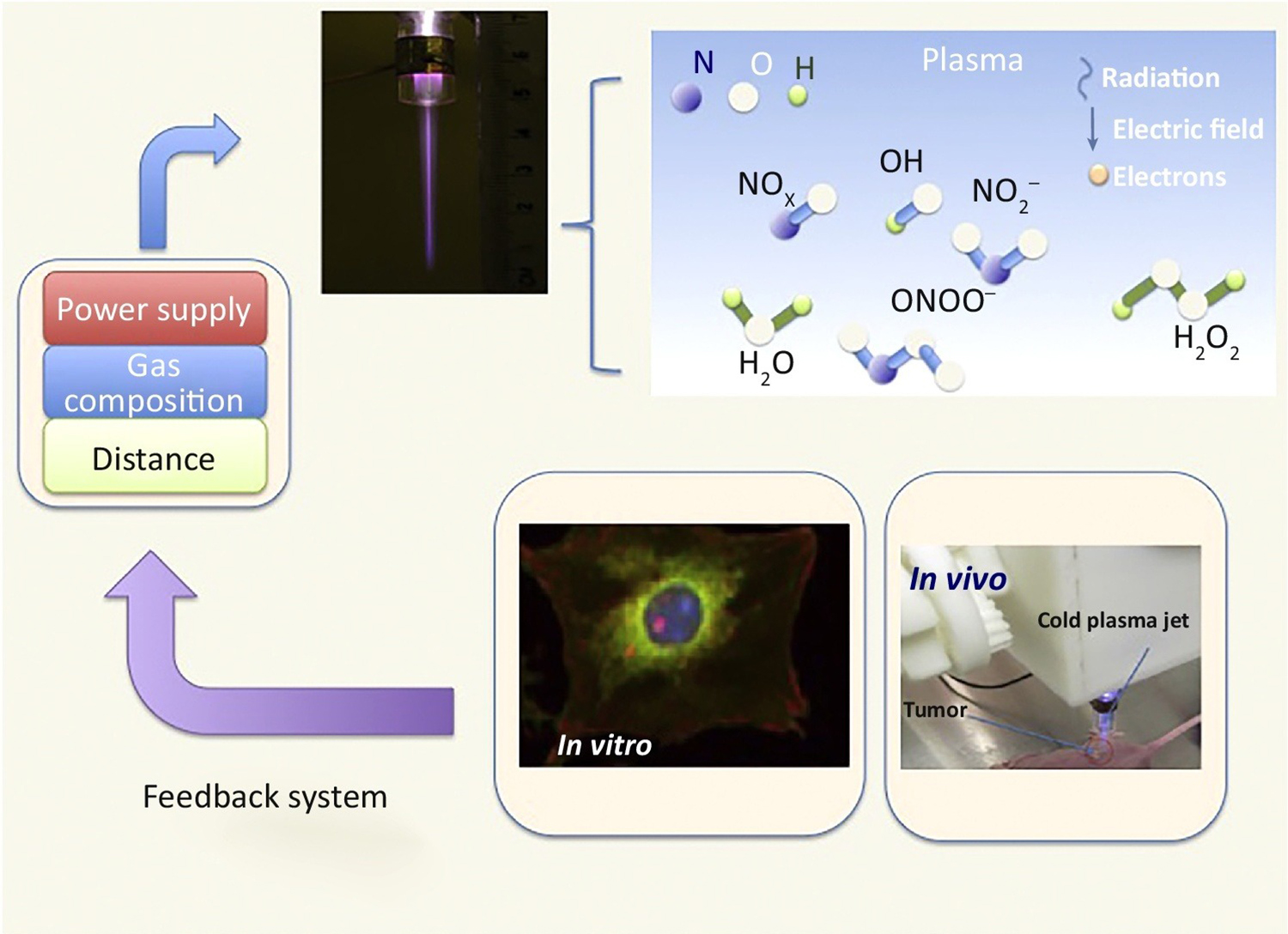
<p style="text-align:justify"><font size ="3"> Plasma self-organization enables adaptive features in plasma-based therapeutic systems. MpNL is investigating the adaptive properties of plasma in order to develop an adaptive CAP system. The novel system will enhance cancer selectivity while relying on a feedback mechanism for controlling levels of CAP-generated reactive species or potential plasma-cell interactions. Publication of a relevant study can be found <a href="http://www.sciencedirect.com/science/article/pii/S0167779917301622"> here</a>.</font></p>
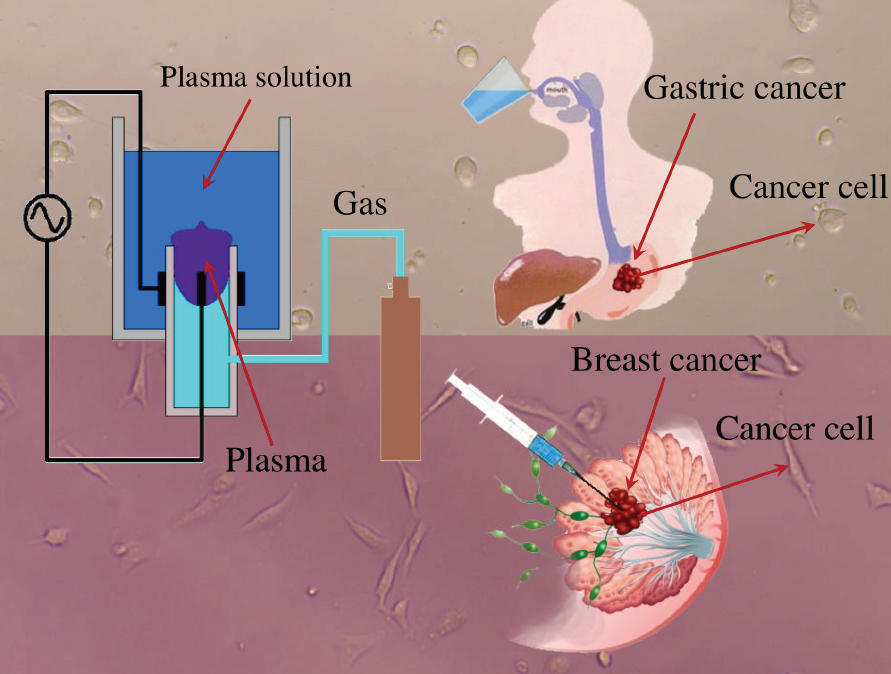
<p style="text-align:justify"><font size="3">MpNL has reported CAP-activated water as a possible non-invasive method for the treatment of gastric and breast cancer. MpNL's research was featured as a cover story in the journal of Plasma Processes and Polymers. The full article can be accessed <a href="http://onlinelibrary.wiley.com/doi/10.1002/ppap.201670036/abstract"> here</a>.</font></p><!-- [et_pb_line_break_holder] -->
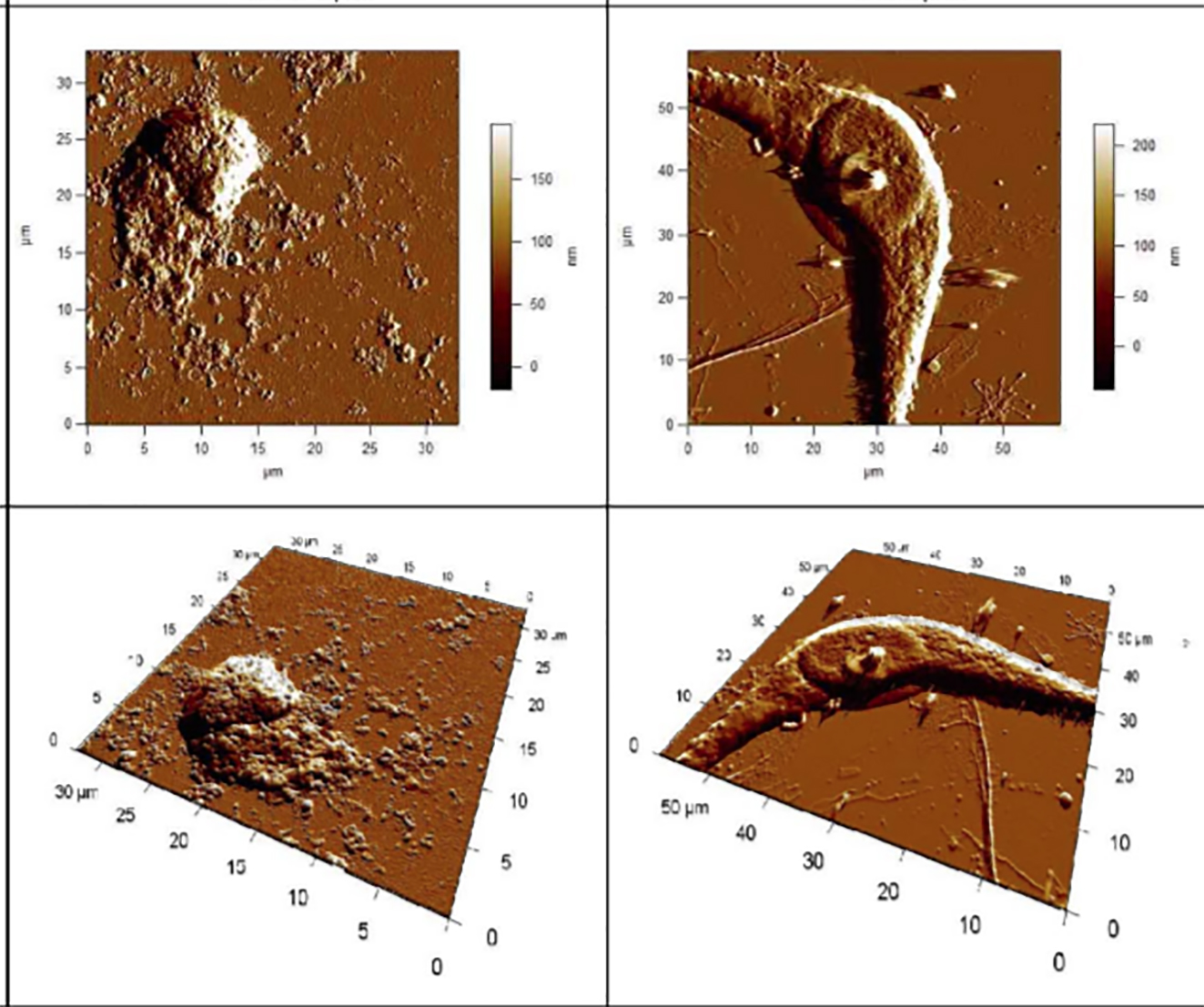
<br><!-- [et_pb_line_break_holder] --><p style="text-align:justify"><font size="3">Gold nanoparticles in combination with CAP can increase glioblastoma cell death by up to thirty percent relative to CAP-only treatment. The article can be accessed <a href="http://iopscience.iop.org/article/10.1088/0022-3727/47/33/335402/meta "> here </a>.</font></p><!-- [et_pb_line_break_holder] -->
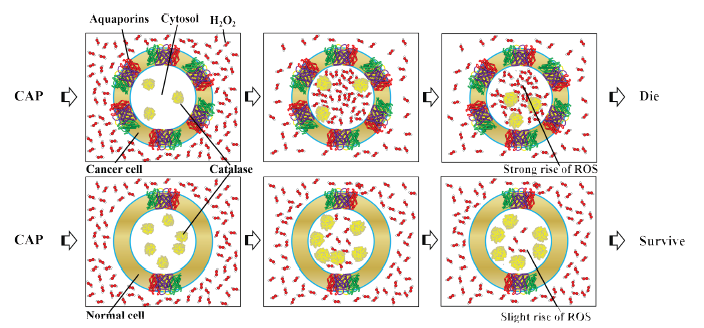
<p style="text-align:justify"><font size ="3">A novel model was proposed based on the expression of aquaporins (AQP) on the cytoplasmic membrane of cancer cells. AQP are integral membrane proteins which serve as channels in the transfer of water and other fluids. They play a key role in tumor growth, angiogeogenesis, and metastatic processes. A recent MpNL study found that cancer cells consume hydrogen peroxide much faster than normal cells due to higher AQP expression.</font></p>

<p style="text-align:justify"><font size ="3">A novel plasma devise, μCAP, was developed for plasma delivery through an intracranial endoscopic tube to target glioblastoma tumors in the mouse brain. Helium µCAP treatment maintained tumor volume below basal levels. While in control condition, tumor volume aggressively increased over time. Publication of this study can be reached <a href="http://www.mdpi.com/2072-6694/9/6/61"> here</a>.</font></p><!-- [et_pb_line_break_holder] -->

<p style="text-align:justify"><font size="3"> A CAP jet was investigated as a potentially transformative cancer treatment modality. A copper ring was used to control the chemical composition of the CAP jet. When a Helium CAP jet goes through the ring, which builds up an electrostatic field, the ring potential increases the electron density and drastically changes the optical emission distribution along the jet. A relatively low potential before the jet can increase the amount of excited N2, N2+, He, and O, but can reduce NO and OH. Publication of this study can be found <a href=" http://aip.scitation.org/doi/10.1063/1.4961924"> here</a>. </font></p>
